CARE1: A European Study on First-Line Treatment of Metastatic Kidney Cancer

CARE1 is a European project coordinated by Gustave Roussy, initiated on May 1st, 2023. Focused on pragmatic clinical trials, it aims to improve the first-line treatment for patients with metastatic kidney cancer by implementing a routine biomarker, leveraging a unique academic network in Europe.
In 2020, Europe saw over 138,000 new cases of clear cell renal carcinoma, the most common form of kidney cancer, resulting in 50,000 deaths (source: GLOBOCAN).
To define research priorities in academia and launch dedicated clinical trials for this tumor, European oncologists have joined forces under the CARE group. This consortium brings together 14 partners, including four from Cancer Core Europe, representing 8 European countries (France, Germany, Spain, the Netherlands, Italy, Austria, the Czech Republic, and the United Kingdom). CARE1 also includes two patient associations. Its goal is to assist oncologists in selecting the best treatment combination for each patient.
Optimizing Treatments for Metastatic Renal Carcinoma
The treatment of clear cell renal carcinoma relies on a combination of two classes of agents: anti-angiogenic targeted therapy (VEGFR TKI - vascular endothelial growth factor receptor tyrosine kinase inhibitor) and immunotherapy (immune checkpoint inhibitors targeting PD-1/PD-L1 or CTLA-4).
However, there has been no direct comparison between these two approaches as first-line treatments, and patients have been treated based on the physician's decision, without clinical or biological factors guiding the treatment choice.
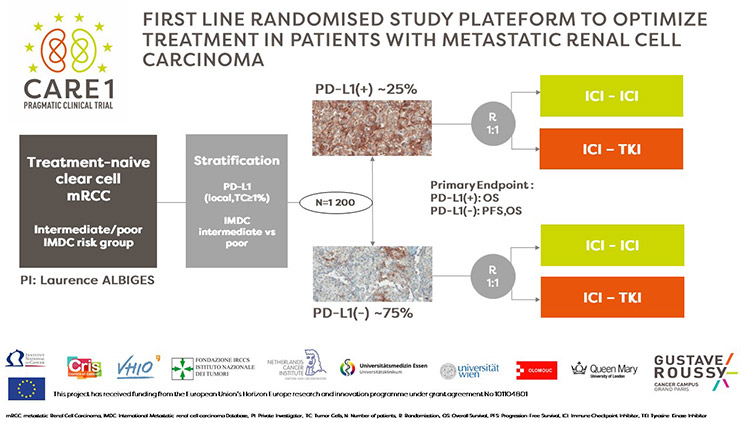
CARE1 was designed to determine the optimal combination for patients using a routine implementable biomarker. This prospective randomized phase III study aims to recruit 1,250 treatment-naive patients with clear cell metastatic renal carcinoma from 8 European countries to compare the combination of two immunotherapies versus the combination of targeted therapy + immunotherapy, based on the tumor's PD-L1 status. PD-L1 status for each patient is determined through immunohistochemistry conducted by the pathology laboratory at the patient's treatment center. If the tumor expresses the PD-L1 marker, it's PD-L1+, otherwise, it's PD-L1-. The primary endpoint of the study is overall survival in the PD-L1+ population, with a co-primary endpoint of overall survival and progression-free survival in the PD-L1- population.
CARE1 will establish a large-scale platform to define the best standard of treatment based on a routine biomarker to enhance treatment efficacy.
In the long term, a data collection effort, including the creation of a pathological and blood biobank, will be conducted on a European scale.
This program is supported by European funding of 5.5 million euros (Grant agreement no. 101104801).
Project Coordinator: Prof. Laurence Albiges, Head of the Department of Oncology.
Partners: Gustave Roussy, Gustave Roussy Transfert, Centre Hospitalier Universitaire de Rennes, Universitatetsklinikum Essen (Germany), Fundacio Privada Institut d’Investigacio Oncollogica de Vall d’Hebron (Spain), Stichting Het Nederlands Kanker Instituut (Netherlands), Servicio Madrileno de Salud (Spain), Fundacion para la Investigacion Biomedical (Spain), Fondazione IRCCS Istituto Nazionale Dei Tumori (Italy), Medizinische Universitaet Wein (Austria), Fakultni Nemocnice Olomouc (Czech Republic), International Kidney Cancer Coalition (Netherlands), RESILIENCE (France), PRIMAA (France), Queen Mary University of London (United Kingdom).

The project CARE1 has received funding from the European Union’s Horizon Europe research and innovation programme under grant agreement
No°: 101104801
